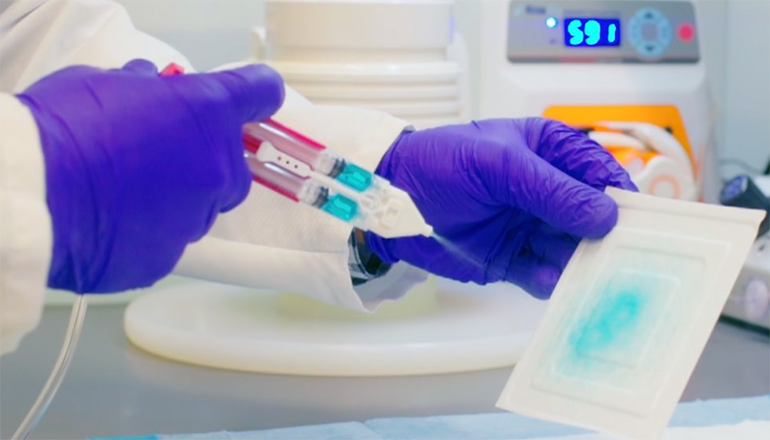
BioDevek, a medical technology company based in Cambridge, Massachusetts, has developed a biomaterial adhesive that seals internal wounds and cuts. The material is designed to be sprayed through a catheter and the main use of the technology to date has been to act as a seal after a colon polyp resection.
Currently, post-polyp resection surgeons can use polyp clips to seal the resection site, which can impede wound healing, or they can use no sealant at all, resulting in an open wound that is associated with the risk of bleeding or other complications. This can lead to additional surgery to correct the problem and is inconvenient for both the patient and the doctor.
To address this, these researchers fine-tuned this biomaterial so that it forms an immediate seal over the site of a resected polyp as soon as it is removed from a catheter. The sealant has been optimized to provide the right mix of mechanical properties and biocompatibility for this application, but can be modified for use on other parts of the body.
BioDevek was recently listed as a finalist in the MedTech Innovator Global 2021 competition.
Watch a video about the company’s technology:
Medgadget had the opportunity to speak to Natalie Artzi, co-founder of BioDevek, about the technology.
Conn Hastings, Medgadget: Please give us an overview of the difficulties encountered in sealing wounds or cuts after internal operations.

Natalie Artzi, BioDevek: Any material that gets into our body is immediately recognized as foreign. Therefore, the formulation of the material is critical in order to achieve the “correct” biological response. Adhesive sealants are naturally designed to chemically and physically interact with tissue surfaces, making it even more difficult to get the correct tissue reaction.
Adhesive sealants must match the mechanical properties of the underlying fabric in order to enable the fabric to function properly. At the same time, the sealant must adhere to the tissue to prevent the suture line from leaking after internal operations.
Commercially available adhesive materials have either high mechanical properties or sufficient biocompatibility that doctors have to choose between the two. BioDevek is working to solve this problem by developing intelligent materials that meet both of these properties. We believe that the development of such materials will increase the penetration of adhesive materials, which has previously been limited due to the sub-optimal performance of existing products.
Medgadget: What are the consequences of sub-optimal wound sealing for patients?
Natalie Artzi: Inadequate wound closure can lead to leaks such as bleeding after vascular surgery, intestinal leakage after gastrointestinal surgery and air leakage after lung surgery in up to 30% of patients who undergo internal operations. This leakage can require additional procedures and even emergency surgery, increasing the risk of possible infection and affecting the overall wound healing time. Eliminating these complications and allowing the body to heal naturally enables shorter hospital stays and lower costs for patients and providers.
Medgadget: What commercial solutions are currently available for such applications? How are they suboptimal?
Natalie Artzi: Apart from sutures and staples, there are currently no commercially available biomaterial-based solutions for wound closure after internal operations. In recent years we have seen a significant need for better wound closure techniques. For example, the frequency of bleeding after polyp resection in the colon has increased. Polyp resection clips are used for polyps smaller than two centimeters, but they can fall off prematurely and hinder wound healing, as they put a lot of mechanical stress on the tissue. For polyps larger than two centimeters, the clips are too small to attach and patients are sent home with an open wound. This puts patients at high risk of delayed bleeding and increases the potential for reoperation and blood transfusion.
Medgadget: Please give us an overview of the sealant developed by BioDevek.

Natalie Artzi: BioDevek’s adhesive sealant is designed to be sprayed through a catheter from one of the existing ports of currently used colonoscopy devices to coat and protect the patient’s tissue after polyp resection. It’s an easy and quick process as the material is designed to gel instantly upon contact with the tissue. Since no two patients are alike, our material is designed to interact with each tissue in a unique way to maximize its performance. We have also developed an injectable solution that can coat the anastomotic line between tissue and graft after cardiovascular surgery.
Medgadget: How long does the seal stay in place? Is it possible to fine-tune the gel degradation and mechanical properties for different uses?

Natalie Artzi: The material is designed to stay in place for about a week after polyp resection, but its residence time can be fine-tuned for each application by controlling its formulation. The breakdown time can be programmed to match the rate at which tissue is healing. In this way, the material provides protection while the tissue is still “compromised” and falls off when the tissue is fully healed.
Medgadget: Was it difficult to develop a formulation that could be administered through a catheter?
Natalie Artzi: Developing the ideal adhesive sealant that can be administered through a catheter has been a learning experience. The sealant dispensing operations must be very quick and take less than a second. The material should have a viscosity low enough to flow easily through a catheter only a few millimeters in diameter, but should form a gel immediately after spraying without flowing or dripping. As the gel solidifies, it must interact with the tissue and at the same time interact with itself to form a highly cohesive solid gel. We are in the process of refining our solution, but we believe we are on the right track to provide an innovative option for patients undergoing internal surgery.
Medgadget: Congratulations on being a finalist in this year’s MedTech Innovator Global competition. How did you like the program?
Natalie Artzi: We are pleased that BioDevek was named a finalist in this year’s global MedTech Innovator competition and look forward to accelerating our development schedule with the prize money we deserve. A really priceless part of this trip was the care we received from corporate sponsors of the program such as Gore & Associates, Olympus Medical Systems, and Jabil Healthcare, among others. These conversations have helped shape the direction of our company for the future. We sincerely enjoyed our time at the Accelerator and look forward to using what we learned from the program to provide better solutions for patients undergoing internal surgery.

Link: BioDevek homepage …